1,2-Dichloroethane (data page) – Wikipedia
From Wikipedia, the free encyclopedia
Chemical data page
This page provides supplementary chemical data on 1,2-dichloroethane.
Structure and properties[edit]
Thermodynamic properties[edit]
| Phase behavior | |
|---|---|
| Triple point | 237.2 K (–35.9 °C), ? Pa |
| Critical point | 561.6 K (288.5 °C), 5380 kPa |
| Std enthalpy change of fusion, ΔfusH |
8.8366 kJ/mol at –35.9 °C |
| Std entropy change of fusion, ΔfusS |
37.25 J/(mol·K) at –35.9 °C |
| Std enthalpy change of vaporization, ΔvapH |
33.91 kJ/mol at 20 °C |
| Std entropy change of vaporization, ΔvapS |
? J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH |
–169.7 kJ/mol |
| Standard molar entropy, S |
208.53 J/(mol K) |
| Enthalpy of combustion, ΔcH |
–1236.4 kJ/mol |
| Heat capacity, cp | 129.0 J/(mol K) |
| Gas properties | |
| Std enthalpy change of formation, ΔfH |
–125.4 kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | 77.5 J/(mol K) at 25 °C |
Vapor pressure of liquid[edit]
| P in mm Hg | 1 | 10 | 40 | 100 | 400 | 760 | 1520 | 3800 | 7600 | 15200 | 30400 | 45600 | |
| T in °C | –44.5(s) | –13.6 | 10.0 | 29.4 | 64.0 | 83.4 | 108.1 | 147.8 | 183.5 | 226.5 | 272.0 | — | |
Table data obtained from CRC Handbook of Chemistry and Physics 44th ed. The (s) annotation indicates temperature is equilibrium of vapor over solid. Otherwise temperature is equilibrium of vapor over liquid.
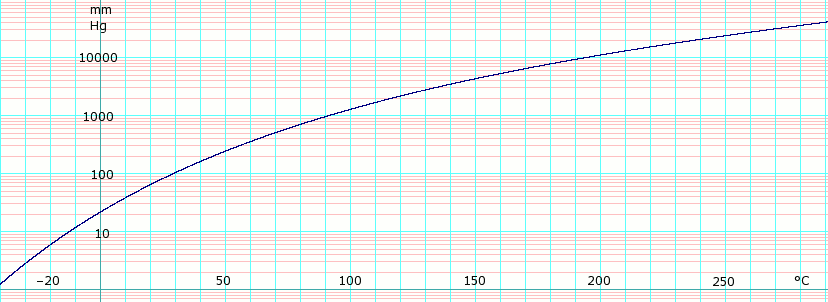
log10 of 1,2-dichloroethane vapor pressure. Uses formula: obtained from CHERIC[5]
Distillation data[edit]
See also:
|
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Spectral data[edit]
References[edit]


Recent Comments